The table below shows the exact mass of each isotope isotopic mass and the percent abundance sometimes called fractional abundance for the primary isotopes of Carbon. Divide the percent abundance from Step 4 by 100 to get the relative abundance.
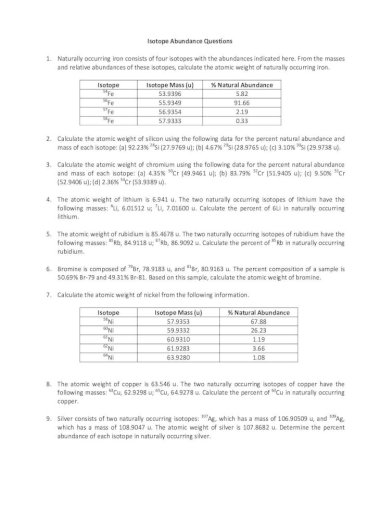
Isotope Abundance Worksheet Abundance Questions 1 Naturally Occurring Iron Consists Of Four Isotopes With The Abundances
How to Calculate Percent Abundance.

. The three Potassium isotopes have atomic masses and percent compositions of 39 amu 648 40 amu 186 and 41 amu 166. You may not need to use all the rows 2. Using the following data calculate the average atomic mass of magnesium give your answer to the nearest 01 u.
Calculating atomic mass worksheet. Silicon-28 9223 2797693 amu Silicon-29 468 2897649 amu Silicon-30 309. 2 Two isotopes of Rubidium occur naturally.
Calculating Average Atomic Mass Worksheet. 1 Three isotopes of Silicon occur in nature. Antimony has two naturally occurring isotopes.
Strontium consists of four isotopes with masses of 84 abundance 050 86 abundance of 99. Ad Download over 20000 K-8 worksheets covering math reading social studies and more. In this worksheet we will practice calculating percentage isotopic abundances from the relative atomic mass and isotopic masses.
Firstly property called distributive is used. The answer 12011 amu is the same value found for. What is the average atomic mass of europium.
Calculate the actual atomic mass of 65Cu. Consider that an element has two isotopes. Silicon-28 9223 2797693 amu Silicon-29 468 2897649 amu Silicon-30 309 2997377 amu Calculate the average atomic mass for the three isotopes of Silicon.
Learning objectives Define atomic mass. Carbon 6 C 12011 isotope abundance mass amu. 6 12C 6 13C.
How to Calculate Percent Abundance. Calculating Relative Abundance in Mass Spectroscopy If a mass spectrum of the element was given the relative percentage isotope abundances are usually presented as a vertical bar graph. This problem demonstrates finding the percent abundance of isotopes with known average atomic mass.
To find the average atomic mass for Carbon. Add the similar terms in the equation to get the final answer. It has a pile of stones to move and needs to decide what equipment you want to rent to move them.
Use the equation in question 1 to calculate the atomic mass of an element that has two isotopes each with 5000 abundance. The percent abundance for Ne-21 is 02700. From these data calculate the.
The problem above is simplified. Calculate the relative atomic mass of strontium. Calculating Average Atomic Mass Worksheet.
12 25Mg Percent abundance. If the stones are small. Find the percent abundance.
Using the average mass from the periodic table calculate the abundance of each isotope. One isotope has a mass of 6300 amu and the other has a mass of 6800 amu. Calculating percent abundance of isotopes worksheet.
The other isotope 65Cu has an abundance of 3091. 65Cu 649278 amu 7 Magnesium consists of three naturally occurring isotopes. Write your final answer for Abundance in the Abundance column.
Then like terms is combined given by. Calculate the relative abundance of the unknown isotope. Naturally occurring boron B consists of two isotopes with a mass of 10 and 11.
Europium-151 has an abundance of 4803 and Europium-153 has an abundance of 5197. Atomic mass mass 1 1 mass 2 2. In this worksheet we will practice calculating percentage isotopic abundances from the relative atomic mass and isotopic masses.
The relative abundances of these four isotopes are 14 241 221 and 524 respectively. 12 24Mg Percent abundance. Finally solving for x we get x076.
Absolute abundances of isotopes are com-monly reported in terms of atom percent. Here are three isotopes of an element. Determining the Mass Due to Isotopes in the Isotope Mass Column.
Given below is a problem with its detailed solution explaining how to find the same. Now we will use the algebra to find the value of x. If the atomic masses of these isotopes are given calculating the percentage abundance of each of the isotopes is possible.
Ne-20 mass 199924 amu Ne-21 mass 209938 amu Ne-22 mass 219914 amu. Isotope Practice Worksheet 1. The abundance of the nitrogen-14 isotope is 996 percent and the abundance of the nitrogen-15 isotope is 04 percent.
Using the Percent Calculation Column in Data Table 2 calculate the percent abundance of each isotope using the equation in the top row of the column. For example atom percent 13C C12C 13C100 1 A closely related term is the fractional abundance fractional abundance of 13C 13F 13F 13C12C 13C 2. Abundance is defined as the amount of isotope contained in its parent element.
Divide the mass of each isotope beans peas and corn by the number of each isotope to get the average mass of each isotope. Calculating percent abundance of isotopes worksheet Next. Naturally occurring europium Eu consists of two isotopes was a mass of 151 and 153.
I find this approach helps students to understand the principle of a weighted average. Find the percentage. 65Cu 305475 Calculation of percent abundance of isotopes is usually done in chemistry worksheets.
Worksheet to help students calculate RAM from relative abundances of isotopes. Here is a neon problem involving all three isotopes. Find the percent abundance for Ne-22.
The relative abundance of 3 7 C l in an average sample of chlorine is 3 7 3 5 C l C l 0. The value of atomic mass of copper is 63546. Calculate the average atomic mass of bromine showing all work.
The mass of antimony-121 is 120904 amu and the mass of antimony-123 is 122904 amu. Structured to start by thinking of balls in a box through to simple calculation for copper. 3 1 9 6.
Calculate the atomic mass given the relevant information on the isotopes. A scaffolded worksheet giving students practise in calculating relative atomic mass from masses of isotopes and percentage abundance. Average Atomic Mass 120000 9890 130033 00110 12011 amu.
Divide the number of each isotope the total number of particles and multiply by 100 to get the percent abundance of each isotope. Average Atomic Mass Worksheet. The average atomic mass between these two isotopes is 63546 amu.
24Mg 7870 25Mg 1013 and 26Mg 117. Chlorine consists of two isotopes with masses of 35 abundance 75 and 37 abundance of 25. Recalculate the atomic mass if instead there is 8000 of the 6300 amu isotope and 2000 of the 6800 amu isotope.
Chlorine has two stable isotopes 3 5 C l and 3 7 C l with atomic masses 349689 u and 369659 u respectively. There are actually three stable isotopes of neon with the unmentioned one being Ne-21. The percent abundance of these isotopes is as follows.
12 26Mg Percent abundance. 1 Three isotopes of Silicon occur in nature. Isotope effects and their consequences in open and closed systems.
Isotopes - relative abundance calculations structured worksheet.











































